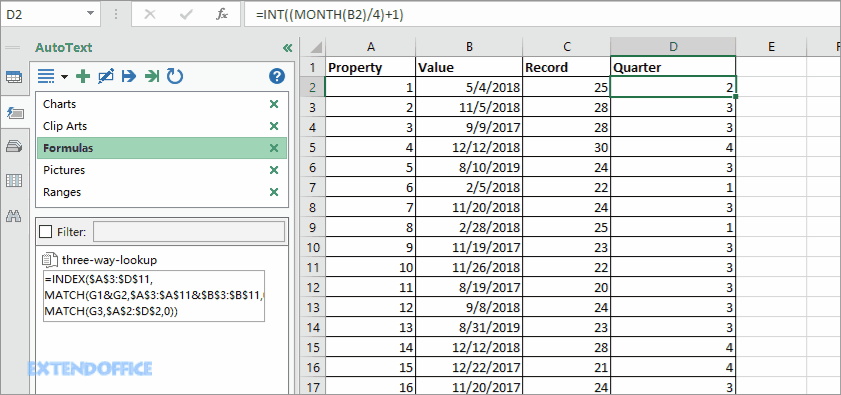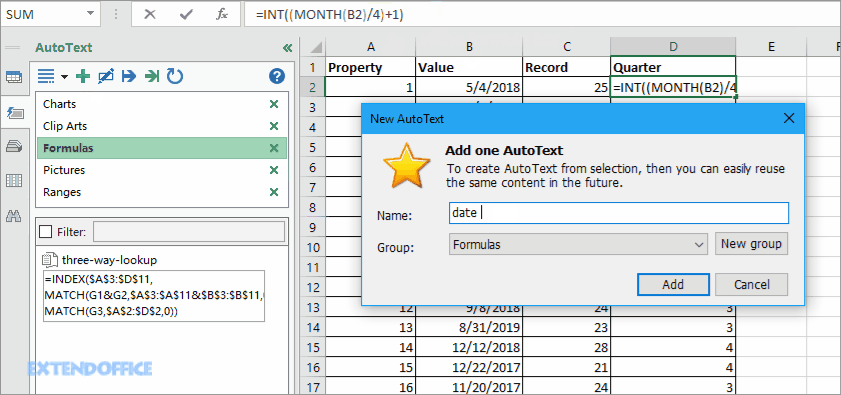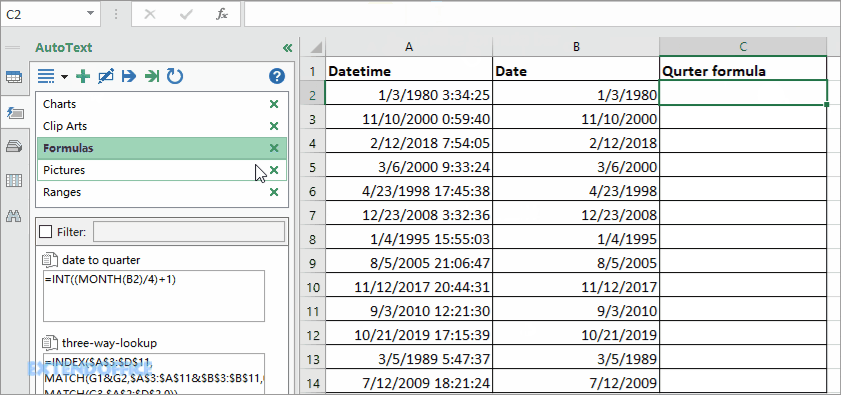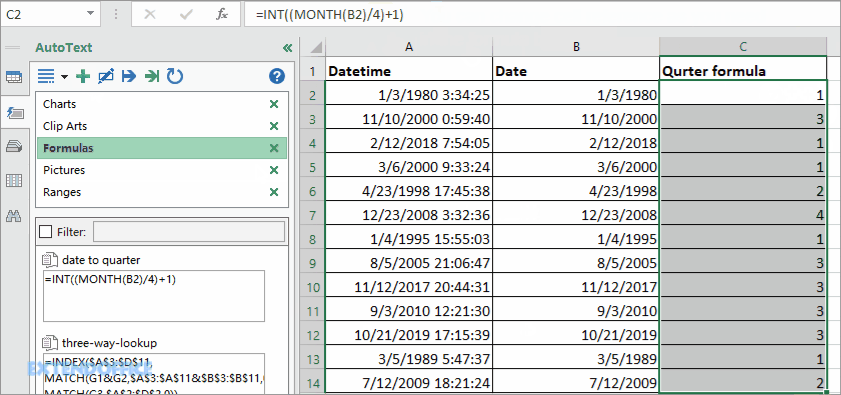
Non-GAAP diluted EPS is expected to be in the range of ($0.44) to ($0.14) per share. The Company expects diluted EPS in the range of ($0.60) to ($0.30) per share, which reflects the Company’s continued investment to advance its whole-product Surmodics expects fiscal year 2020 revenue to range from $87 million to $91 million. Capital expenditures totaled $1.9 million for the fourth quarter of Surmodics generated $12.5 million of cash from operating activities in the fourth quarter of fiscal 2019.

On a non-GAAP basis, earnings per share were $0.37 in the fourth quarter of fiscal 2019, asĬompared with $0.05 in the year-ago period.Īs of September 30, 2019, cash and investments were $55.3 million. In Vitro Diagnostics revenue was $6.0 millionįor the fourth quarter of fiscal 2019 as compared with $6.1 million in the same prior-year quarter, a decrease of 1%.ĭiluted GAAP earnings per share in the fourth quarter of fiscal 2019 were $0.26 as compared with a loss per share of $(0.13) in the year-ago period. Revenue in the fourth quarter includes $7.6 million from our SurVeil agreement with Abbott, as compared with $2.2 million in the prior-year quarter.

Medical Device revenue was $24.8 million in the fourth quarter of fiscal 2019, as compared with $17.0 million in Total revenue for the fourth quarter of fiscal 2019 was $30.8 million, as compared with $23.0 million in the prior-year period. I am excited by our achievements to date and look forward to continuedįourth Quarter Fiscal 2019 Financial Results Unpredictability related to the Paclitaxel debate, our team has demonstrated impressive execution which included completing enrollment for our TRANSCEND clinical trial. “During fiscal 2019, we made important progress across our key strategic objectives while delivering strong organic revenue and solid operational results,” said Gary Maharaj, President and CEO of Surmodics, Inc. Signed agreement with a leading multi-national medical device partner for worldwide distribution of Surmodics’.Submitted application for first in-human study for Sundance™ below-the-knee DCB granted “Breakthrough Device Designation” from the FDA.Completed enrollment in first in-human study for Avess™ arteriovenous (AV) access DCB.
#Srdx qtr date trial#
Completed enrollment in TRANSCEND pivotal clinical trial for SurVeil™ drug-coated balloon (DCB).GAAP EPS of $0.26, non-GAAP EPS of $0.37.Revenue of $30.8 million, an increase of 33.8% year-over-year.Summary of Fourth Quarter and Recent Highlights Quarter ended September 30, 2019, and provided its financial outlook for fiscal 2020. (Nasdaq: SRDX), a leading provider of medical device and in vitro diagnostic technologies to the healthcare industry, today announced results for its fiscal 2019 fourth EDEN PRAIRIE, Minn.-(BUSINESS WIRE)-October 31, 2019-Surmodics, Inc.
